 
We write \(S^o_A\left(T\right)\) to indicate the absolute entropy of substance \(A\) in its standard state at temperature \(T\). It is usually included in compilations of thermodynamic data for chemical substances. The standard entropy is usually given the symbol \(S^o\). 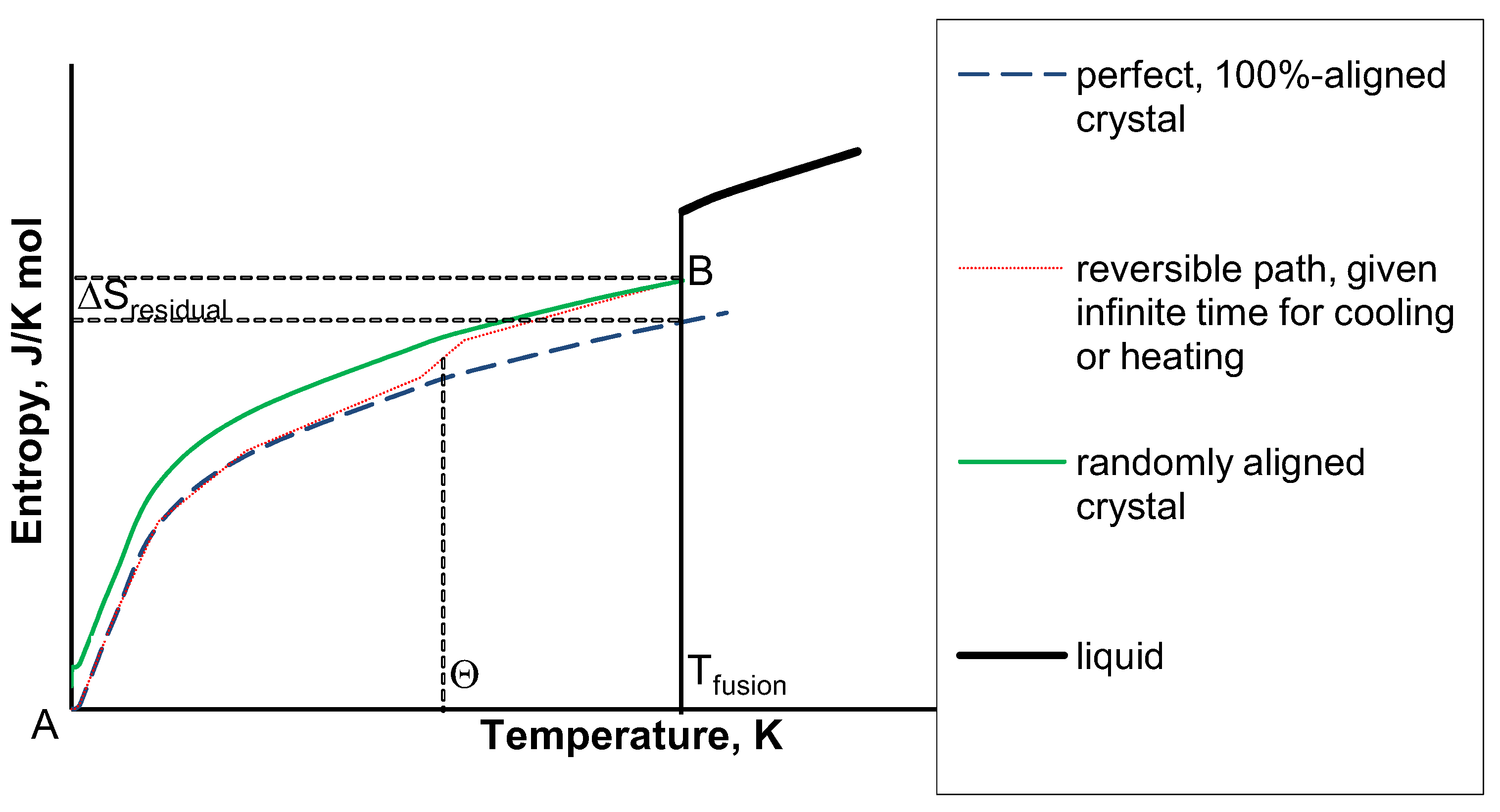
When the entropy value is calculated for one mole of the substance in its standard state, the resulting absolute entropy is called the standard entropy. Such conclusions are derived from the measurements of the heat capacity of the substance. Where the substance undergoes phase changes, the contribution that the phase change makes to the entropy of the substance is equal to the enthalpy change for the phase change divided by the temperature at which it occurs.Īt any given temperature, the entropy value that is obtained in this way is called the substance’s absolute entropy or its third-law entropy. The results suggest that the RRHO&MIE strategy could be useful for estimating absolute and/or relative entropies of single molecules either in the gas phase. One of the prime applications of the third law of thermodynamics is that it supports calculating the absolute entropy at any particular temperature (T). 1: Standard Molar Entropy Values of Selected Substances at 25C. 2, which is a generalized plot of the entropy of a substance versus temperature. Phase changes are isothermal and reversible. The correlation between physical state and absolute entropy is illustrated in Figure 9.3.2 9.3. In temperature ranges where experimental heat capacity data are available, the entropy change is obtained by integration using these data. Compute answers using Wolframs breakthrough technology & knowledgebase, relied on by millions of students & professionals. The entropy contribution due to lattice vibrations. Natural Language Math Input Extended Keyboard Examples Upload Random. For example, if the temperature is constant: f-i Nln Vf-Nln Vi Nln(Vf/Vi) 1 2 V V1 2. On collecting the different contributions to the excess entropy of mixing, we obtain S xs S xs,conf + S vib + S el + S mag Sex,conf has been discussed in the previous section and the relevant expression is given in Eq. Entropy of an ideal gas: For N distinguishable particles in volume V: VN Nln V+ const You can’t calculate the constant (that requires quantum mechanics), but it drops out of problems where one only needs the entropy change. \), using Debye’s theoretical relationship, \(C_P=AT^3\) \(A\) is obtained from the value of \(C_P\) at the lowest temperature for which an experimental value of \(C_P\) is available. Simple scheme for the excess entropy of mixing. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
